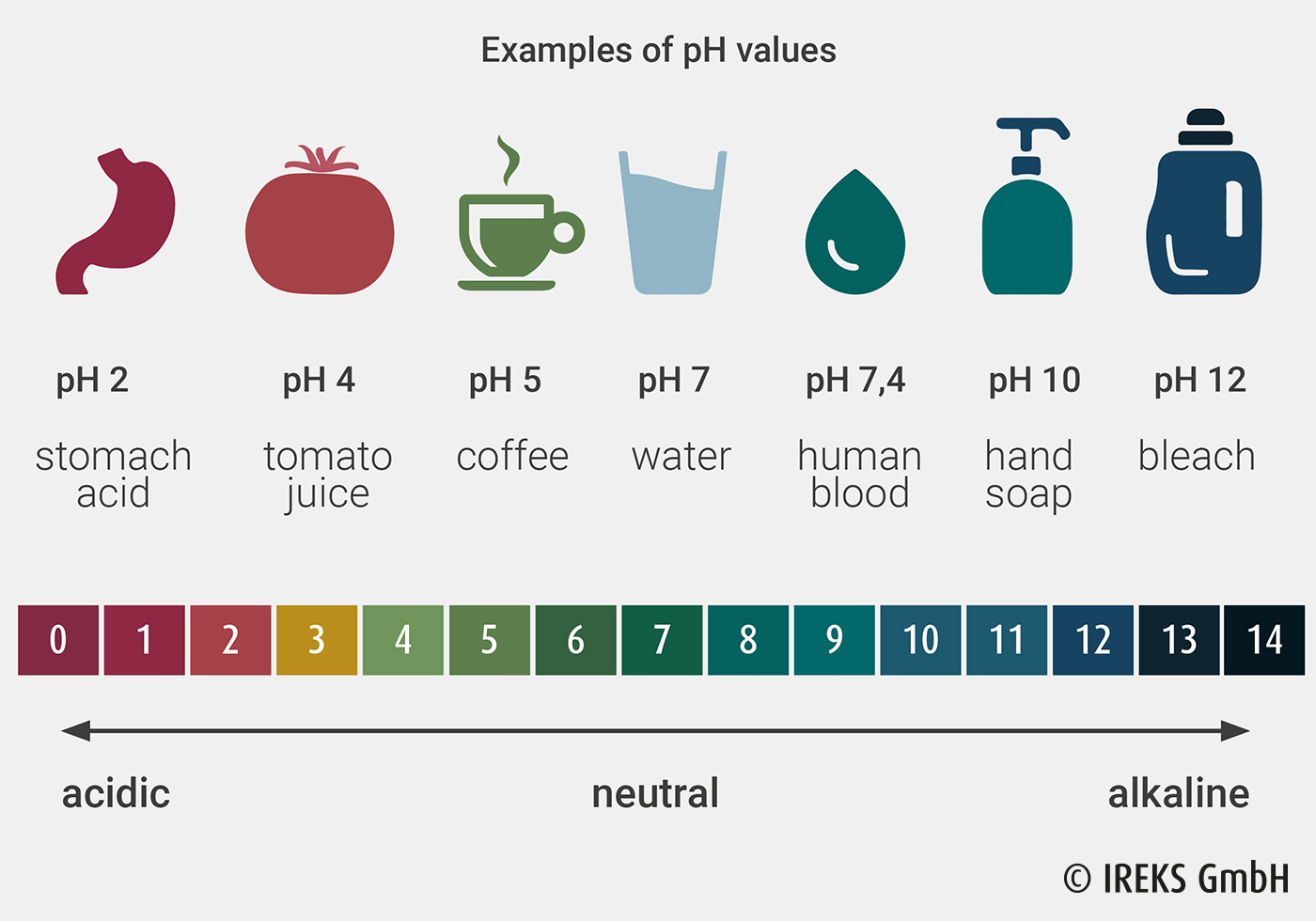
The pH value is the index for the effective acid or base in a diluted solution. Here, the pH value corresponds to the negative decadic logarithm of the hydrogen ion concentration or, more exactly, the hydrogen ion activity. Here, the pH value range reaches from 0 sour via 7 neutral to 14 alkaline (Illustration 15.8). The pH value shows the strength of the acid or alkaline base here.
In addition to the pH value, the degree of acidity can be determined. The degree of acidity is the measure of the total quantity of acid in the product. The sodium hydroxide (0.1 mol/l) used, which is usually required to neutralize an aqueous suspension of 10 g substance (e.g. dough, bread or sourdough), is measured.
By means of the pH value and degree of acidity, conclusions can be drawn regarding the maturity of the sourdough and the correct acidification of the bread. More information on the pH value and degree of acidity are contained in the Chapter Rye sourdoughs.